- Energy & Environment
- Science & Technology
In his provocative new book “Zero to One,” Silicon Valley venture capitalist Peter Thiel states that America’s economic success depends on making “vertical, or intensive,” advances, rather than “horizontal, or extensive,” progress. While horizontal progress is having one typewriter and manufacturing a hundred, vertical progress is going from a typewriter to a word processor. As Mr. Thiel puts it, vertical progress “means doing new things—going from 0 to 1” with technology.
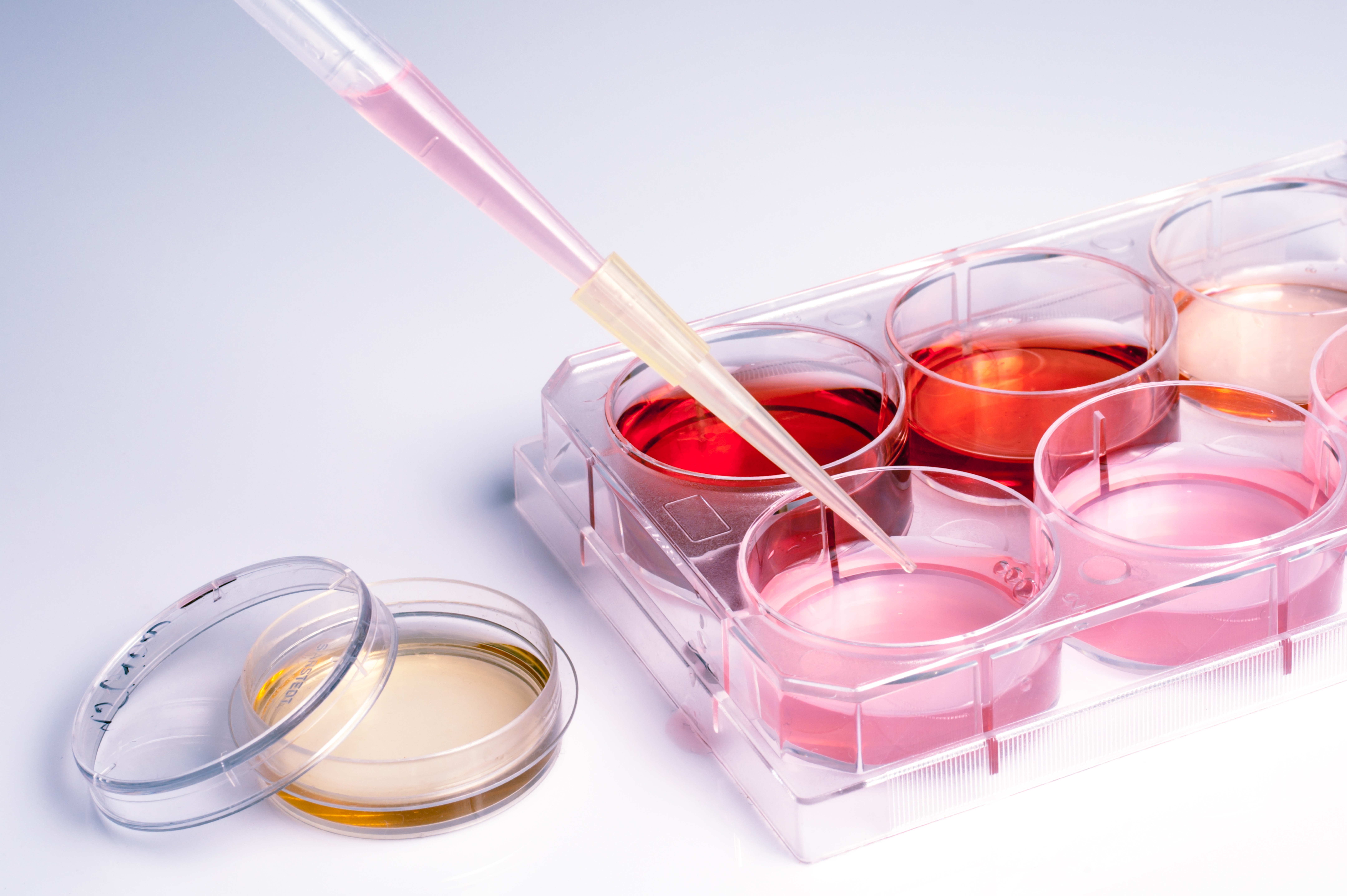
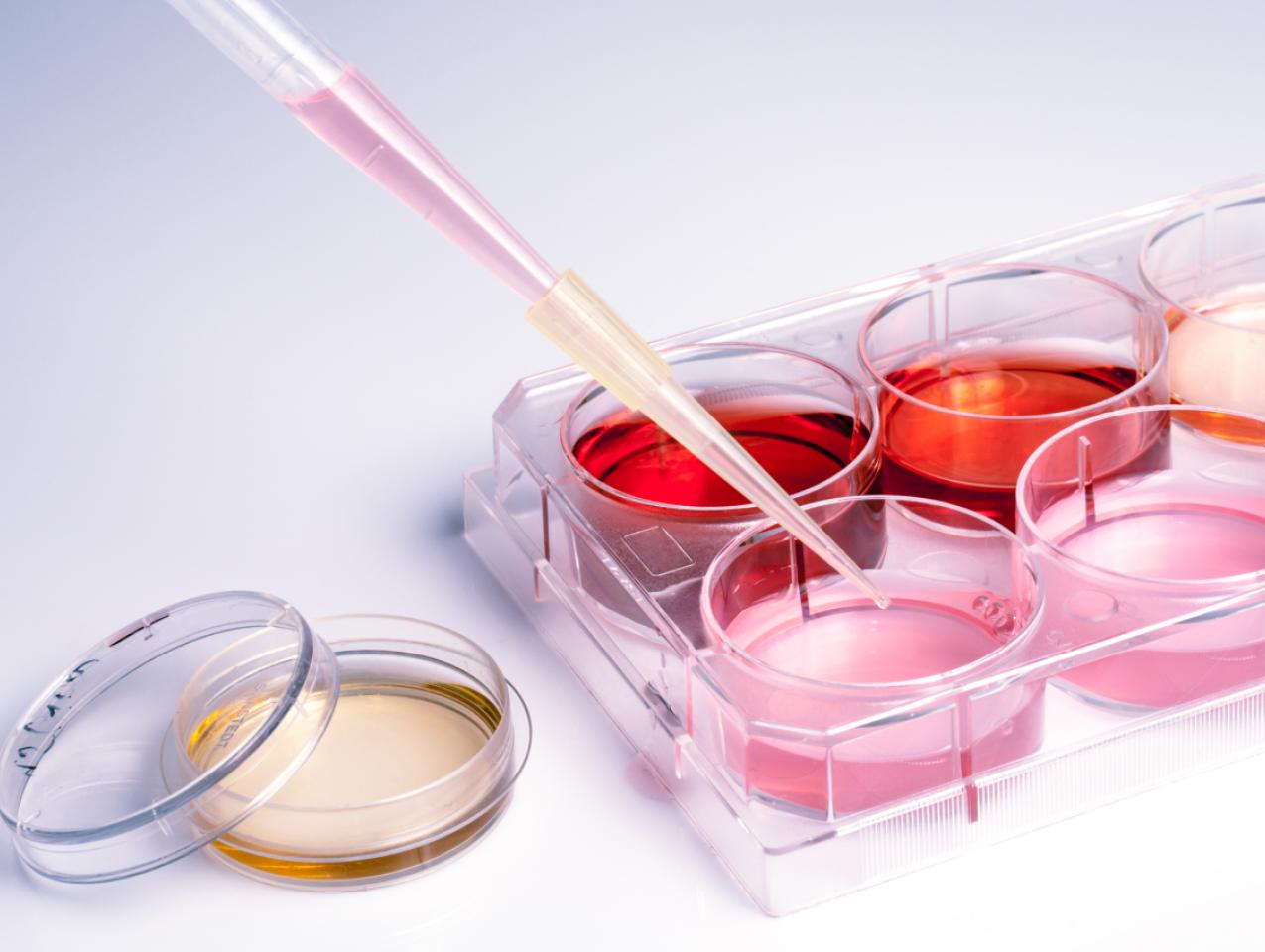
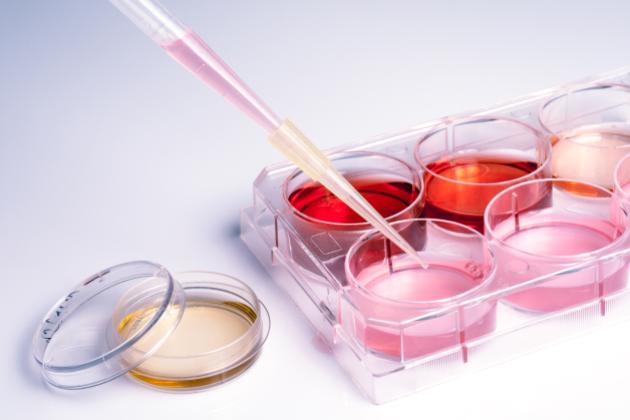
Modern genetic engineering, also called genetic modification or GM, has been around since the 1970s. Yet with the notable exception of biopharmaceuticals—beginning with the marketing of human insulin in 1982 and now accounting for more than 20% of U.S. drug expenditures—genetic engineering has failed to realize anything approaching its potential for vertical progress.
The reason is plain: In the non-pharmaceutical sectors, federal regulators for years seemingly have done everything they can to prevent U.S. researchers and companies from employing genetic engineering to create the “next big thing.”
Early on the federal government tried to take the right approach. In 1986 the White House Office of Science and Technology Policy (OSTP) published a policy statement on genetic engineering that focused oversight and regulatory triggers on risk-related characteristics, such as plants’ weediness or toxicity. The policy rejected regulation based on the particular process used for genetic modification, since genetic modification has been used for centuries through a variety of techniques—such as selective breeding and hybridization, including “wide crosses,” in which genetic material is moved from one species or genus to another.
Six years later OSTP reaffirmed that the degree and intrusiveness of oversight “should not turn on the fact that an organism has been modified by a particular process or technique.” These statements reflected a long-standing consensus among scientists that the newer techniques of genetic modification are essentially an extension, or refinement, of older, less-precise and less-predictable ones and that oversight should focus on the characteristics of products, not on the processes or technologies that produced them.
But the Environmental Protection Agency and the Department of Agriculture had other ideas. They created new regulations that focus on recombinant DNA, or gene-splicing, technology, which is among the most precise and predictable techniques. But they exempted others, such as wide-cross hybridization and mutagenesis by irradiation or chemicals.
This approach ignores that the particular genetic technique employed to construct new strains is irrelevant to risk, as is the source of a snippet of DNA that may be moved from one organism to another. What matters are the traits that are related to risk, such as allergenicity or the presence of toxins, not the use of one technique or another. Unfortunately, federal regulators have consistently chosen to focus on the latter.
Regulatory disincentives are potent. It costs about $136 million to bring a genetically engineered crop plant to market. This is the primary reason more than 99% of such crop plants are those that are grown at huge scale: Corn, cotton, canola, soy, alfalfa and sugar beets. Hawaiian papaya is a rare example of significant acreage of genetically engineered “specialty crops” such as fruits, nuts and vegetables.
“Biopharming”—the once-promising biotechnology area that uses genetic engineering techniques to induce crops such as corn, tomatoes and tobacco to produce high concentrations of high-value pharmaceuticals (one of which is the Ebola drug, ZMapp)—is moribund because of the Agriculture Department’s extraordinary regulatory burdens. Thanks to EPA’s policies, which discriminate against organisms modified with the most precise and predictable techniques, the high hopes for genetically engineered “biorational” microbial pesticides and microorganisms to clean up toxic wastes have evaporated.
As a result, the potential for vertical progress—for innovation—that modern genetic engineering holds for long-term, robust U.S. economic growth and higher living standards has been drastically reduced. Amazon CEO Jeff Bezos also made this point in the context of developing commercial drones at a conference in December. “Technology is not going to be the long pole,” he said. “The long pole is going to be regulatory.”
In the absence of scientifically defensible regulation, innovation will suffer, and the U.S. will forgo who knows how many big things.
Dr. Miller, a physician and molecular biologist, is a research fellow at Stanford University’s Hoover Institution. He was the founding director of the FDA’s Office of Biotechnology.